Prices incl. VAT plus shipping costs
In stock, delivery time approx. 1-2 days**
- Order number: ASTMF3039BPIP1
- EAN 4251496617326
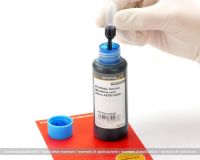
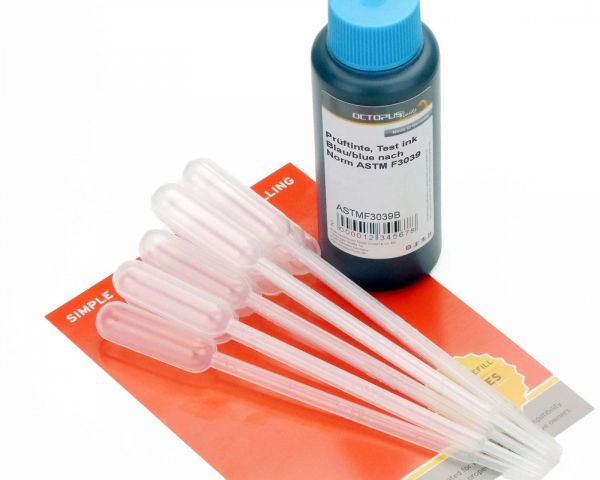
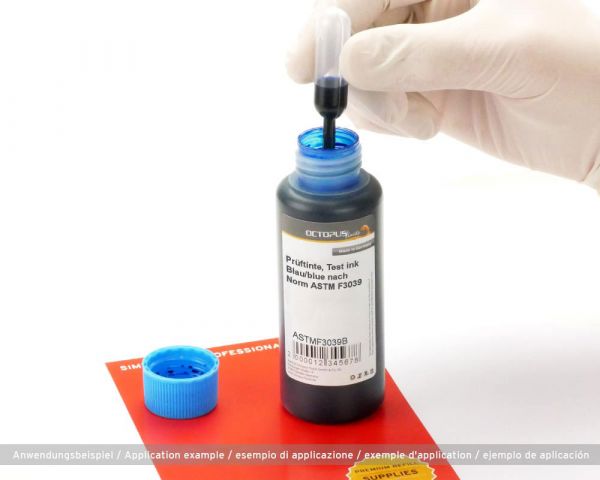
This blue test ink complies with the current norm ASTM F3039 for testing packaging for medical devices to be sterilized in the final packaging. The test ink is available in stable HDPE plastic bottles with 10 pipettes. The ink has a minimum shelf life of 2 years. This test ink is also available in other colours on request and with the corresponding purchase quantity.
Norm ASTM F3039: “Packaging for terminally sterilized medical devices - Part 1: Requirements for materials, sterile barrier systems and packaging systems”
Note: The ASTM F3039 standard applies to non-porous sealing materials (film-film pouches made of PE, PET, PVA, PVOH, PVC). When using partially porous sealing materials (paper-film pouches) the test ink according to DIN EN ISO 11607-1, Annex B (in American ASTM F1929) is required.
Norm ASTM F3039 specifies a test procedure that can be used to determine whether sealing seams of medical packaging seal sterilely. Also known as seal tightness test. During the functional test of sealed seams of medical packaging, this ink is used to check the seam tightness. If the ink leaks, it can be assumed that the medical device is not sterile and the packaging process should be adapted.
These test inks according to norm ASTM F3039 are manufactured by Octopus Fluids GmbH & Co. KG and their physical properties are adjusted according to norm ASTM F3039.
When testing sealing seams, our test ink makes constant and reliable statements thanks to the consistent quality of our batch test. The ink is also available in further colours. For this you may make your inquiry without obligation.
| Content: | 500 ml |
| Minimum storage life: | 2 years |
 Test ink blue according to DIN EN ISO 11607-1, ASTM 1929 with pipettes
Test ink blue according to DIN EN ISO 11607-1, ASTM 1929 with pipettes
 Transfer pipettes with dosage tip and plastic bulb, vol. 1 ml
Transfer pipettes with dosage tip and plastic bulb, vol. 1 ml
 Test ink blue according to DIN EN ISO 11607-1, ASTM 1929 with pipettes
Test ink blue according to DIN EN ISO 11607-1, ASTM 1929 with pipettes
 Transfer pipettes with dosage tip and plastic bulb, vol. 1 ml
Transfer pipettes with dosage tip and plastic bulb, vol. 1 ml







 Manufacturer info
Manufacturer info